Are your patients using the latest technology?
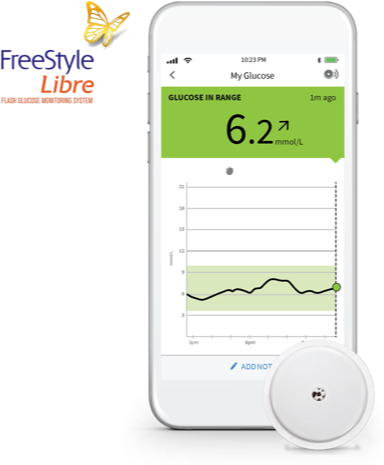
The FreeStyle Libre 2 system gives your patients the knowledge to effectively manage their diabetes and improve their glycemic outcomes.1,2*
Are your patients using the latest technology?
The FreeStyle Libre 2 system gives your patients the knowledge to effectively manage their diabetes and improve their glycemic outcomes.1,2*
Sensors stick better when patients wash, clean and dry their skin before application.
The FreeStyle Libre digital network keeps everyone in the know.
Lets parents and caregivers easily monitor their loved ones’ glucose readings from their phone‡‡
The FreeStyle Libre system is covered by Ontario,*** Quebec,††† Yukon‡‡‡, PEI§§§ and the NIHB¶¶¶ public reimbursement programs for eligible patients.
The FreeStyle Libre flash glucose monitoring system is indicated for measuring interstitial fluid glucose levels in adults aged 18 years and older with diabetes mellitus.4
The FreeStyle Libre 2 system has all of the FreeStyle Libre system’s features, with the addition of three optional, real-time glucose alarms that are available through the FreeStyle Libre 2 app.‡‡‡‡ There is a Low Glucose Alarm, High Glucose Alarm and Signal Loss alarm.
The FreeStyle Libre 2 system is indicated for measuring interstitial fluid glucose levels in people aged 4 years and older with diabetes mellitus.5
No, FreeStyle Libre system sensors and the FreeStyle LibreLink app** are NOT compatible with FreeStyle Libre 2 system sensors, readers or the FreeStyle Libre 2 app.†††† It is critical that patients use the matching sensors and app to ensure product functionality. The FreeStyle LibreLink app** is available through the App Store or Google Play.
Most private health insurance companies also cover the FreeStyle Libre system. Encourage your patients to ask their private insurance provider if the FreeStyle Libre system is covered for them.
The target glucose range is pre-set to 3.9-10 mmol/L. Patients can adjust their target glucose range via the Settings menu.
Follow these guidelines when travelling:4
Check with the airline prior to departure as rules and regulations may change without notice. without notice.
Patients should remove the sensor prior to MRI, CT scan or X-ray. Patients should apply a new sensor after the appointment.4
The FreeStyle LibreLink app** lets users easily share their glucose data with their caregivers using the LibreLinkUp app.‡‡
When caregivers use the LibreLinkUp app,‡‡ they will get glucose readings sent to their phone from their loved one using the FreeStyle LibreLink app.** Every FreeStyle LibreLink scan is automatically¶¶ sent to their smartphone, and up to 20 people can view results from a single FreeStyle LibreLink** account.
For more information and support, encourage patients to visit LibreLinkUp.com.
* Data collected from the FreeStyle Libre system. FreeStyle Libre 2 has the same features as the FreeStyle Libre system, with the addition of optional real-time glucose alarms. Study data are applicable to both products.
† Based on the number of users worldwide for the FreeStyle Libre systems compared to the number of users for other leading personal-use, sensor-based glucose monitoring systems.
‡ The reader can capture data from the sensor when it is within 4 cm of the sensor.
§ Sensor is water-resistant in up to 1 metre (3 feet) of water. Do not immerse longer than 30 minutes. Not to be used above 10,000 feet.
¶ To get a complete glycemic picture over the last 3 months, the sensor must be replaced every 14 days and the sensor must be scanned at least once every 8 hours.
** The FreeStyle LibreLink app and the FreeStyle Libre reader have similar but not identical features. A finger prick test using a blood glucose meter is required during times of rapidly changing glucose levels when interstitial fluid glucose levels may not accurately reflect blood glucose levels or if hypoglycemia or impending hypoglycemia is reported by the FreeStyle LibreLink app or when symptoms do not match the app readings. The FreeStyle Libre sensor communicates with the FreeStyle Libre reader that started it or the FreeStyle LibreLink app that started it. A sensor started by the FreeStyle Libre reader will also communicate with the FreeStyle LibreLink app. The FreeStyle LibreLink app is only compatible with certain mobile devices and operating systems. Please check the Compatibility Guide (PDF 69 KB) for more information about device compatibility before using the app. Use of LibreLinkUp and the FreeStyle Libre 2 app may require registration with LibreView.
†† The LibreView website is only compatible with certain operating systems and browsers. Please check www.libreview.com for additional information.
‡‡ The LibreLinkUp app is only compatible with certain mobile devices and operating systems. Please check www.librelinkup.com for more information about device compatibility before using the app. Use of LibreLinkUp and the FreeStyle Libre 2 app may require registration with LibreView. The LibreLinkUp mobile app is not intended to be a primary glucose monitor: home users must consult their primary device(s) and consult a healthcare professional before making any medical interpretation and therapy adjustments from the information provided by the app.
§§ Treatment decisions should not be based on real-time sensor glucose readings alone but instead should consider all the information on the results screen.
¶¶ Automatic upload requires a wireless internet connection or mobile data connection.
*** For people managing diabetes with insulin.
††† For residents of Quebec, the following two criteria must be met: intensive insulin therapy (insulin pump treatment or ≥3 insulin injections per day), and frequent hypoglycemic events during the last year.
‡‡‡ For people over 18 years of age with type 1 diabetes.
§§§ Those eligible for reimbursement in Prince Edward Island must be 18 years of age or older, be taking three or more daily injections of insulin or using an insulin pump, and must be enrolled in the Diabetes Glucose Sensor Program.
¶¶¶ FreeStyle Libre system (reader and sensors) is a limited use benefit for individuals who are currently managing their diabetes with insulin.
**** Individual private drug plans can vary. Please have your patients check with their plan administrator and/or insurance company.
†††† The FreeStyle Libre 2 app and the FreeStyle Libre 2 reader have similar but not identical features. Finger pricks are required if glucose readings and alarms do not match symptoms or expectations. The FreeStyle Libre 2 sensor communicates with the FreeStyle Libre 2 reader that started it or the FreeStyle Libre 2 app that started it. The FreeStyle Libre 2 app is only compatible with certain mobile devices and operating systems. Please check the Compatibility Guide (PDF 69 KB) for more information about device compatibility before using the app. Use of FreeStyle Libre 2 may require registration with LibreView.
References:
This site is intended for Canadian Healthcare Professionals only. Visit patient site.
The FreeStyle Libre 2 flash glucose monitoring system is indicated for measuring interstitial fluid glucose levels in people aged 4 years and older with diabetes mellitus. Always read and follow the label/insert.
The FreeStyle Libre 3 Flash Glucose Monitoring System (FreeStyle Libre 3 Reader or FreeStyle Libre 3 app used with FreeStyle Libre 3 Plus Sensor) is indicated for measuring interstitial fluid glucose levels continuously in people aged 2 years and older with diabetes mellitus. Always read and follow the label/insert.
The sensor housing, FreeStyle, Libre, and related brand marks are marks of Abbott. mylife and YpsoPump are registered trademarks of Ypsomed AG in several countries. CamAPS is a registered trademark of CamDiab Ltd. Other trademarks are the property of their respective owners. FreeStyle Libre 3 Plus sensors are authorised to work with mylife Loop system, including the mylife CamAPS FX app and mylife YpsoPump insulin pump. For use of the FreeStyle Libre 3 Plus sensors with the mylife Loop, refer to the labeling provided with the mylife CamAPS FX app.
You are now being redirected to a third-party website not under the control of Abbott. Abbott is not responsible for the contents of such site or any further links from such site. Abbott is providing these links to you only as a convenience, and the inclusion of any link does not imply the endorsement of the linked site by Abbott.
Do you wish to leave this site?
EasytoFreeStyle.ca is a product-specific website intended only for residents of Canada.
If you are a resident of another country, please visit our global directory to locate the appropriate site for your country.